 The Pfizer, Moderna and AstraZeneca vaccines are given as two shots in the upper arm muscle, three or. 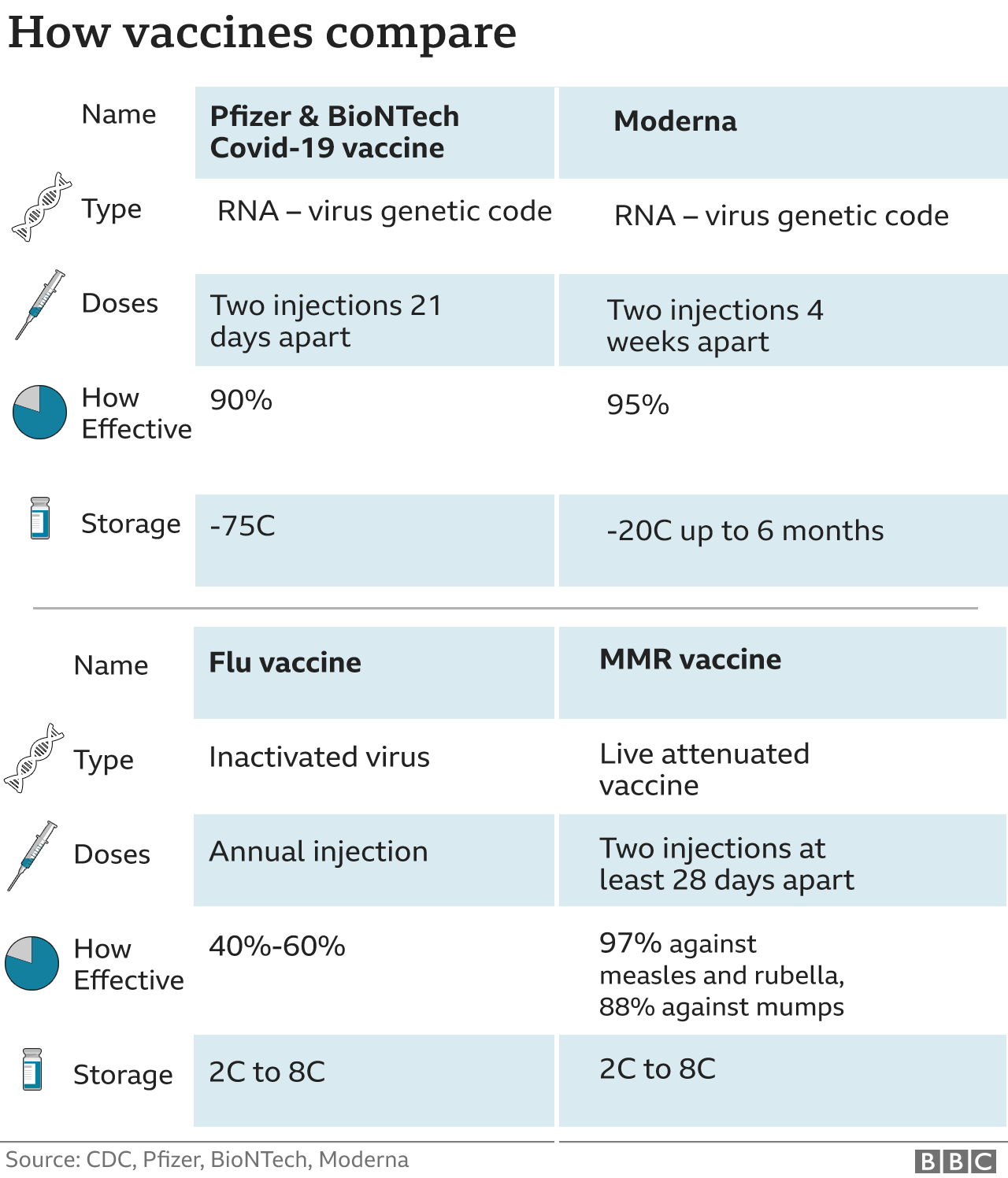 
The company said it is continuing to work to develop an omicron-specific booster. Icon showing vaccine being administered in patients arm. However, these data showing that the currently authorized Moderna COVID-19 booster can boost neutralizing antibody levels 37-fold higher than pre-boost levels are reassuring," said Stéphane Bancel, CEO of Moderna. "The dramatic increase in COVID-19 cases from the Omicron variant is concerning to all. The news of Moderna's findings adds more scientific backing to the idea that booster shots are an effective way to offer continued protection against the coronavirus, especially with the growing omicron variant. Research also showed a 100-microgram booster dose gave an even higher level of protection - an 83-fold increase in neutralizing antibody levels, Moderna said. Moderna COVID-19 Vaccine, Bivalent is authorized for use in individuals 6 years of age and older who were previously unvaccinated or vaccinated with one or more doses of an approved or. The pharmaceutical company said its current FDA-authorized, 50-microgram booster shot increases antibody levels against omicron 37-fold compared to pre-booster levels. Our scientifically trained team are able to combine up-to-date knowledge on the evolving pandemic, with decades of marketing experience to meet you needs.Take a look at our services.Moderna said Monday that preliminary data showed that its COVID-19 booster shot is effective against the omicron variant of the coronavirus currently surging in the U.S. This article provides a summary of those interim recommendations you may access the full guidance document here. We have continued to support clients through the current pandemic, producing technical literature, articles and digital marketing support relating specifically to COVID-19 for the medical, healthcare and biotechnology industries. The WHO Strategic Advisory Group of Experts on Immunization (SAGE) has issued updated interim recommendations for the use of the Moderna COVID-19 (mRNA-1273) vaccine against COVID-19. ** This figure varies dependent on the source and trials in Brazil (50%), UAE (86%) and from the manufacturer (79%) state differing efficacies. * Sputnik V consists of two adenovirus vector constituents (rAd5 and rAd26), Name: BBIBP-CorV, Sinopharm CNBG’s Covid-19 vaccineĬompany press release: China grants conditional market approval for Sinopharm CNBG’s COVID-19 VaccineĬompany press release: Sinovac Announces Phase III Results of Its COVID-19 Vaccine Type: Adenovirus vectorĬompany press release: Johnson & Johnson Announces Single-Shot Janssen COVID-19 Vaccine Candidate Met Primary Endpoints in Interim Analysis of its Phase 3 ENSEMBLE TrialĬompany: CanSino Biologics and Academy of Military Medical Sciences, CNĬompany press release: NMPA Accepts the Application for Conditional Marketing Authorization of CanSinoBIO’s COVID-19 Vaccine, Convidecia TM Type: Protein subunit (spike protein)Ĭompany Press Release: Novavax COVID-19 Vaccine Demonstrates 89.3% Efficacy in UK Phase 3 Trial Type: Inactivated whole virusĬompany press release: COVAXIN® – India’s First Indigenous COVID-19 Vaccine The following vaccines are awaiting peer-review, so alternative sources for efficacy figures, some of which are press releases of interim analysis from the manufacturer, are shown. Peer review awaited: Phase 3 vaccine trials doi:10.1016/S0140-6736(20)32661-1įor further information, our in-depth post on the results of the AZD1222 vaccine trial, covers more detail. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. 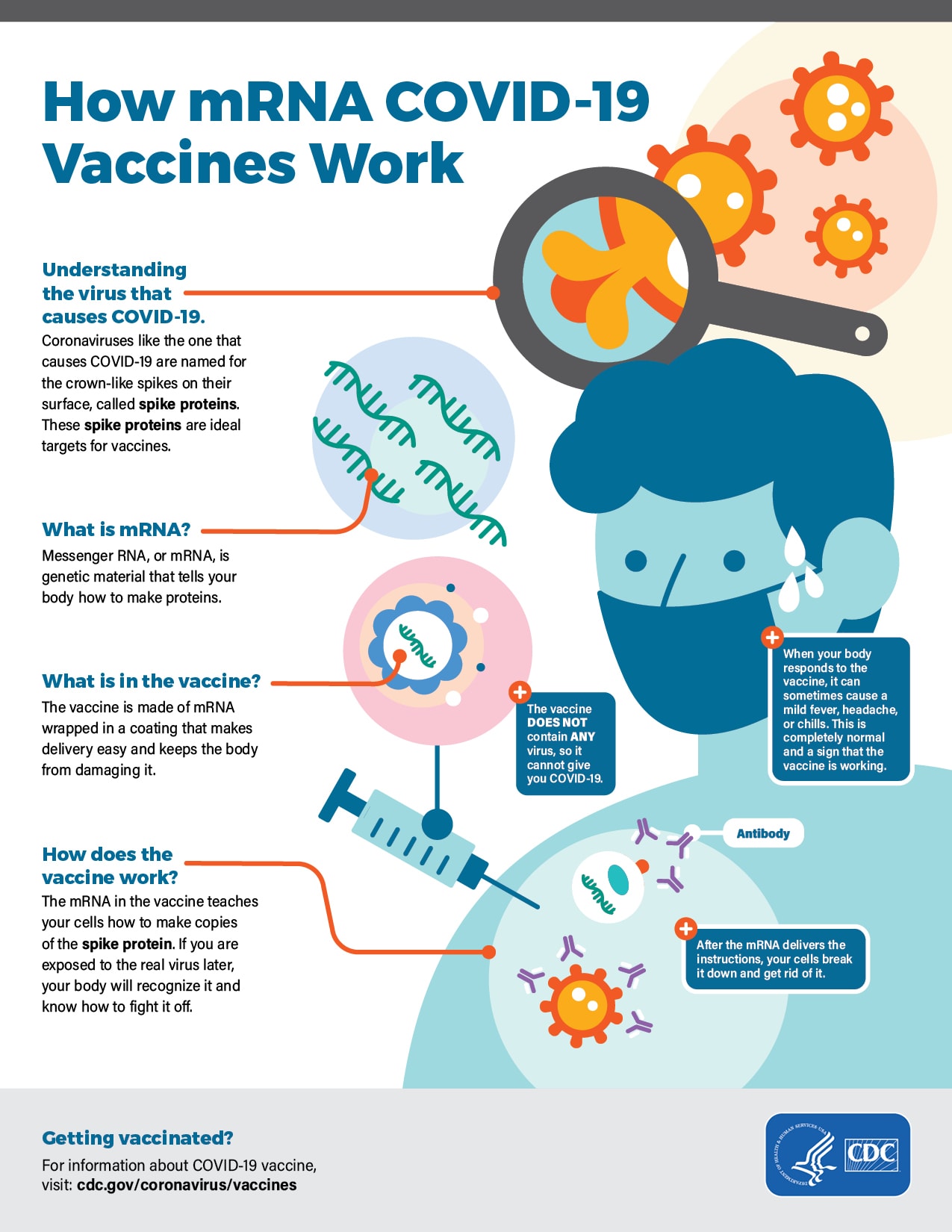
Logunov DY, Dolzhikova IV, Shcheblyakov DV, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. Moderna COVID-19 Vaccine, Bivalent (Original and Omicron BA.4/BA. Name: Comirnaty, BNT162b2 (Pfizer vaccine) Peer Review: Phase 3 vaccine trials Type: mRNA Sign up to get a free PDF version of our covid-19 vaccine summary infographic 
Links to the original reference, where the Phase 3 trial has been published and referenced are shown below. Our vaccine infographic highlights the key Covid-19 vaccines in full or early use, globally. Covid-19 Vaccines: Global landscape of Phase 3 Trials Children aged 6 months to 5 years may need multiple doses of COVID-19 vaccine to be up to date, including at least 1 updated dose of Pfizer or Moderna. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
